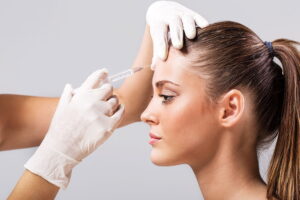
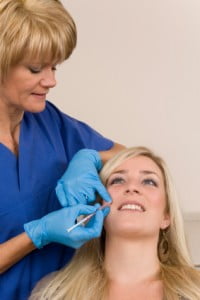
Allergan Inc announced FDA approval of Juvederm XC last week, the latest filler in the U.S. to contain the anesthetic lidocaine, for increased comfort during injection.
During the FDA approval process, a clinical trial was cited where 93 percent of the 72 participants reported a more comfortable procedure with Juvederm XC. “Before the introduction of Juvederm XC, it often took up to 30 minutes for an anesthetic block to take effect.” said Charles Boyd, MD, Boyd Cosmetic Surgical Institute and clinical investigator in a company news release.
The new filler follows in the footsteps of Prevelle Silk, another wrinkle filler containing lidocaine. In 2008 however, when Prevelle Silk was introduced the response from surgeons was lukewarm. “The issue with a hyaluronic acid filler with lidocaine is that it doesn’t work until you actually start injecting patients, and these injections are so quick, by the time the lidocaine takes effect, you’re almost finished” said Dr. Joe Niamtu to Cosmetic Surgery Times.
Traditional pain control methods (numbing cream and dental blocks) are quite effective, so it may take some time for Juvederm XC to gain popularity, even if it does offer a significant improvement in pain control.
The results cited by Allergan sound great and the safety profile is very similar, so it could really take off. We’ll know more as Juvederm XC finds its way to cosmetic surgery practices here in southern California.